
Secure by Demand: Key takeaways for enterprise software buyers
CISA has released an add-on to its supply chain security initiative Secure by Design, with a focus on commercial software. Here’s what you need to know.
Read More











Get the best of RL Blog delivered to your in-box weekly to stay up to date on key trends, analysis and best practices across threat intelligence and software supply chain security.

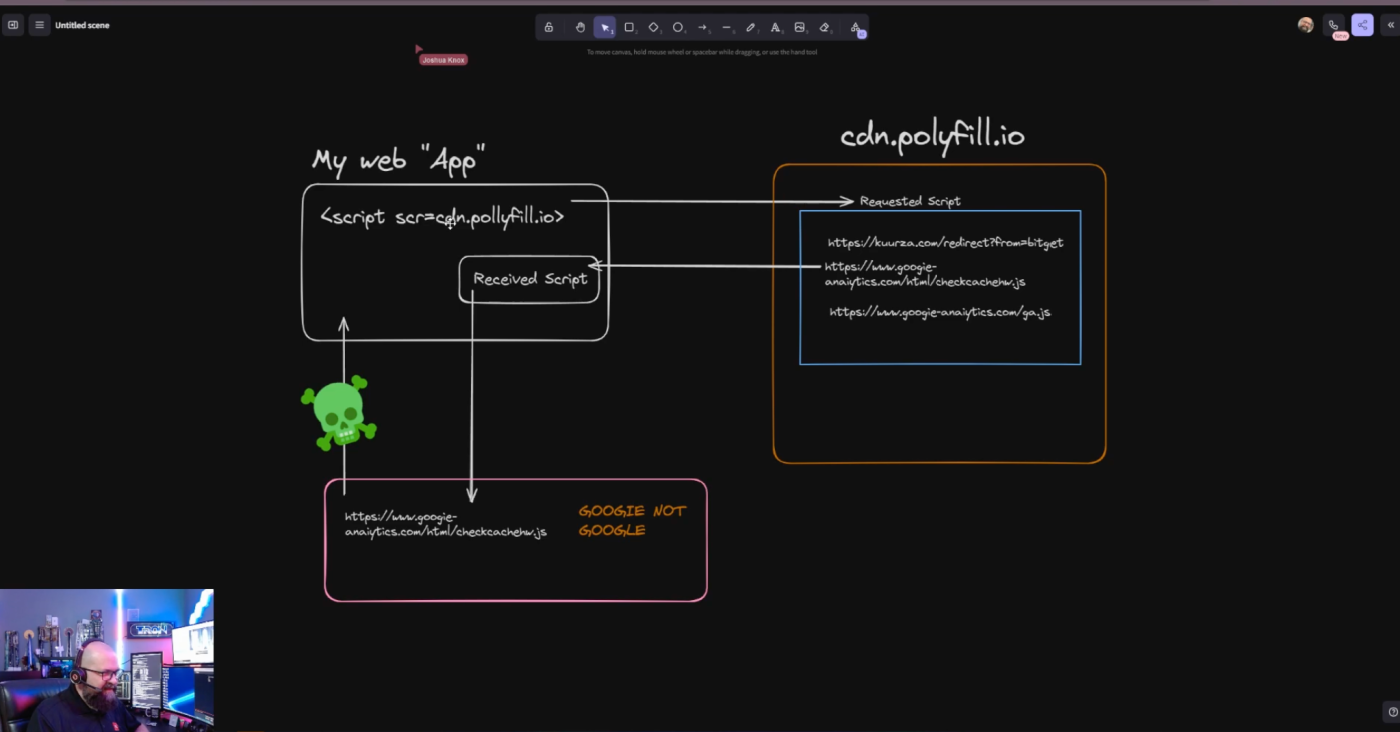
Software supply chain attacks are an increasingly popular tool for malicious actors. And the rapid embrace of AI and machine learning (ML) tools is introducing new supply chain risks. Here's what your organization needs to know.